The Shellfish Gene
How shellfish farmers are leaning on genes to unmask the secrets of mussels, scallops, and oysters.
Article body copy
Four young researchers clamber from a boat onto a teetering raft, sending ripples across calm water that gleams like polished chrome in the October morning sun. The raft is the size of a king-sized bed and all of the students wear life jackets strapped over bulky wool sweaters or bright jackets. Katie Pocock and Tamara Russell maneuver a winch over a hole in the middle of the raft. They throw their weight onto the crank and haul up the first of several cages that hang in the cerulean water. Kayla Mohns cuts the zip ties that were holding the cage closed, revealing the captives within: hundreds of shiny blue-black mussels, frilly blushing scallops, and gnarly Pacific oysters.
While Pocock, Russell, and Mohns get busy selecting samples to take back to the lab, the fourth researcher, Caitlin Smith, is setting up an impromptu wet lab in the damp cabin of the boat moored alongside the raft. She sets out a box of syringes and a rack of test tubes, and picks up a mussel the size of a key fob. She’s extracting hemocytes from the mussel, or as she says, “pretty much mussel blood.” She saws a notch in the mussel with a metal file, then uncaps a syringe and slides the needle into the gap, feeling for the hemocyte-rich adductor muscle. Then, her hands steady and careful as the boat gently rocks, she draws back the plunger, and the barrel fills with clear fluid, chock-full of RNA. Jackpot. She squirts the sample into a tube, caps it, and moves on to the next mussel.
Smith wants RNA because this “messenger molecule” is a snapshot of which genes are being expressed in the animal at that moment. “DNA is just kinda like what codes for the organism, where the RNA is showing how the organism is reacting to its environment,” Smith says. If the mussel is hot or hungry or horny, that message can be found floating in the needle’s barrel.
Cages of shellfish dangle beneath a raft just off the coast of Quadra Island, helping researchers understand the impact climate change is having on these bellwether species. Video by Grant Callegari
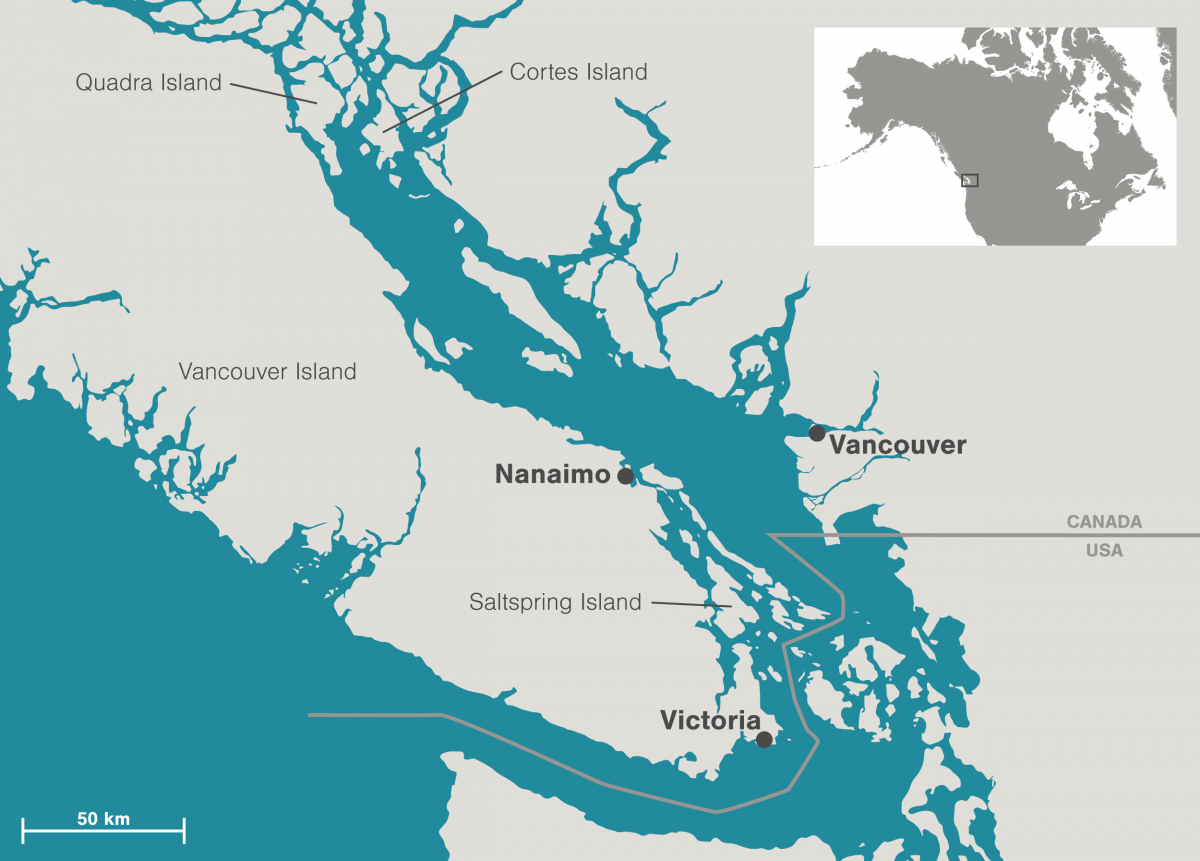
On that October day two years ago, the research assistants were working with Helen Gurney-Smith, then a research scientist at Vancouver Island University and the Hakai Institute.* Now at St. Andrews Biological Station in eastern Canada, her work on the project continues. As part of Gurney-Smith’s quest to understand how shellfish, especially commercial species, are responding to climate change, she set up the raft, the only in situ experiment of its kind on shellfish. The specimens have been dangling for the last two years off the rocky shore of Quadra Island, roughly 150 kilometers northwest of Nanaimo, British Columbia. Above that bay, the Coast Mountains arc in the distance, and blue-green water and verdant forests shake hands along the coast. The conditions below the waves are much harder to see.
Of all the threats to shellfish health, ocean acidification gets the most attention. We humans like a simple bad guy, because it suggests a simple solution. But climate change is a Cerberus, and the answers are not simple. How will shellfish fare in a real, living ocean where change is a constant? Where the water is sometimes too corrosive, sometimes too warm? Where there are increasing outbreaks of toxic algae and the timing of spring plankton blooms keeps shifting? With each change, the effects on shellfish become harder to understand or predict. And shellfish are notoriously inscrutable; they can’t tell you where it hurts.
But their genes can.
Gurney-Smith is conducting her one-of-a-kind experiment to understand how the expression of shellfish genes track with real-time oceanographic monitoring data. The obvious acidic enemy is a key consideration, though she’s also going beyond it to understand a more subtle, and perhaps ultimately more harmful, enemy: chronic, grinding stress from all sides.
Acidification—the result of excess carbon dioxide from the atmosphere getting absorbed into the water—is particularly problematic at the beginning of a shellfish’s life. Larvae extract molecules of calcium carbonate from the water, precipitating them into a mineral (aragonite) to build their shells. Too much CO2 thwarts that effort, turning the water corrosive. While acidification evokes shells bubbling like pennies in cola, the reality is less dramatic; acidic conditions simply force the larvae to work harder to create a shell. That kind of trouble often leads to a thin or deformed shell, which hobbles the animal for life—if it survives.
Once they’re past that critical early stage of shell development, shellfish contend with a whole other host of challenges, including some that are site specific. For instance, shellfish on North America’s west coast contend with CO2-rich, naturally acidic water that has circulated on the ocean bottom and upwells to the surface. The same phenomenon occurs along various coasts, including northern Europe and western Africa. Off the British Columbia coast, the carbon that wells up from the ocean bottom to the surface was actually absorbed from the air 50 years ago—at the moment, shellfish in this part of the world are wrestling with carbon that was emitted during the Cold War. Even if we stopped air pollution tomorrow, carbon from prior emissions would keep upwelling for decades. This means acidification here is hard to predict and is likely to come in pulses rather than rise steadily. Add to that disease, warming, and ecosystem shifts, and you get chronically stressed shellfish.
For shellfish farmers, the uncertainty is a financial stress: in the US Pacific Northwest, the industry is worth US $270-million and in British Columbia, $42-million.
The raft with the oysters, scallops, and mussels is only 50 meters from the wooded shore, where what looks like a small garden shed squats. A dark pipe snakes down to the sea and keeps water collected from just beneath the raft flowing through monitoring equipment inside. This setup belongs to Wiley Evans, a chemical oceanographer with the Hakai Institute and a self-described “CO2 geek.” The system measures dissolved carbon, as well as temperature and salinity, every second, streaming the results online. The data allows Gurney-Smith an unprecedented peek at how shellfish react to the slightest change in their environment.

This raft off the shore of British Columbia’s Quadra Island is home to scallops, oysters, and mussels that can help researchers understand the impacts of climate change on shellfish. Photo by Josh Silberg
Quadra Island, in the relatively sheltered waters of the Salish Sea, isn’t directly exposed to the main coastal upwelling system. Instead, the right wind conditions will periodically bring cold, CO2-rich water flushing down the Strait of Georgia, interrupting the balmy summer conditions. In the space of a week, the CO2 can spike from low summer levels up to higher than winter levels, and back down again, accompanied by temperature drops of up to 8 °C. “You’re basically bathing these animals for a short period of time in corrosive water,” says Evans.
Over several years, Gurney-Smith zeroed in on a suite of known genes—53 in all—that can indicate how shellfish are reacting to environmental stressors. She then tracked how those genes responded to environmental changes using Evans’s real-time data. “There’s a lot of information streaming out of there, and we can use that as an indicator of when we should go up and sample,” she says from her office in New Brunswick. “We’re trying to work out ways to work with organisms that don’t speak, [so] that they can tell us what they’re experiencing.”
Gurney-Smith is now sequencing most of the functional genomes of the shellfish, spotting the activation of previously unknown genes as her shellfish respond to changing conditions. Combined, the data reveals a complex picture of what daily life is really like for the shellfish. Gurney-Smith shares this information with shellfish farmers, which helps them decide which species to invest in, which conditions to monitor, and where to locate their sites for optimal conditions. Gurney-Smith’s genomics have helped one shellfish grower create his breeding lines for the future.
This short video introduces the research raft off Quadra Island and some of the experiments that will tell scientists more about shellfish in a changing ocean.
In a bright, humid lab within a cluster of oatmeal-colored buildings on Saltspring Island, shellfish grower Grant Hunt spreads his hands along the rim of a broad, shallow water basin. “Every other Wednesday, we spawn,” he says. Behind him, a stainless steel tank with a hatched lid burbles. A peek inside reveals racks of adult mussels waiting in the dark. They are, as Hunt says, “ripe.” In the wild ocean, they would stay ripe—aquaculture parlance for horny—until spring, when the water warms and algae blooms. Then they would spew their sperm or eggs into the water to mingle and fertilize. Hunt has kept these prospective parents at 8 °C, which is slightly too cool to mate. Tomorrow, he’ll put them on the table, wash them with warmer salt water, and add some food (phytoplankton), tricking them into thinking it’s springtime.
As soon as a pinhead-sized mussel larva has formed, it must scramble for survival. Before it knows how to eat or what a solid surface feels like, it spends its first 48 hours burning through the shredded remnants of its egg while it builds the foundations of the shell that will house it for life. If conditions are good, the shell is strong and the future is bright. “The shells are like concrete,” Hunt says, “Once you make it, it’s going to last. But it needs the right ingredients.”
Hunt, a zooplankton specialist turned mussel hatchery operator, runs Island Sea Farms’ mussel hatchery on this island 50 kilometers southwest of Vancouver, that’s known for its population of hippies and rich recluses. Hunt is neither. He’s more of an entrepreneur, having designed and built this facility from the ground up. It has the production-line efficiency of a Toyota factory, and the opulence of a five-star resort—for mussels.
Gurney-Smith helped Hunt begin his quest for the perfect hybrids of two local commercial mussel species, Mytilus galloprovincialis and Mytilus edulis (colloquially termed gallo and edulis after their Latin names). Gallos are big, fast growers, but have a thin shell and have trouble with the cold Pacific waters. Edulis are smaller, but they have thicker shells and are hardy. If he merged them together, Hunt thought, he could have a perfect mussel. But first he needed to know what he was starting with, and that required some genetic sleuthing. Gurney-Smith and her team tested Hunt’s broodstock to see whether the mussels he was starting with were pure gallo and edulis. Once he knew which ones were pure, he then created hundreds of different random crosses of each pure strain. He set the resulting larvae out in the water off Cortes Island, a short ferry hop from Quadra Island’s north end and about 200 kilometers from his hatchery. Two years later, Hunt went back to the raft and hand selected the progeny that were doing best: the quick growers, the thick shelled, the well colored. Back at the hatchery, it’s these lines of gallo and edulis mussels that are crossed with each other, producing tough, fast-growing hybrids for the main farm in the waters off Cortes Island.
The hatchery on Saltspring Island draws seawater for its operations up from Trincomali Channel, where tests show the water is mussel nirvana. Because Hunt can monitor incoming flow, he can correct for temperature, acidification, and contamination. Hunt is a farmer of the future, using genetics, climate change science, and good old-fashioned selective breeding. But not all hatcheries or larvae are as pampered, and once the larvae are set out to grow on the rafts, they will need to face the ocean in all its changing moods.

Scallops await dissection at a Vancouver Island University lab. Researchers are studying shellfish, especially commercial species, to understand how they are responding to climate change. Photo by Grant Callegari
The junior members of the lab have moved on to other projects or organizations, but their research has lasting implications for shellfish science. Mohns compared how each of the three species responds to change; for example, how a scallop handles a spike in temperature compared to an oyster. This may help the region’s shellfish growers select species to invest in as conditions change. Smith looked at genes specifically related to immune response and how their expression changes as conditions fluctuate. Russell continues to focus her research on changes in the shellfish’s food: zooplankton and phytoplankton.
Back at a lab in Nanaimo, on a chilly January day, Kayla Mohns reaches into an even-chillier freezer for one of the dozens of frost-furred boxes stacked inside. This one holds scallop gill tissue collected from the raft. If its earlier dissection yielded gourmet castoffs, now the preparation of the specimens resembles following a recipe. Mohns works with the concentration of a master chef, mixing the right chemicals, macerating the concoction to mush in a homogenizer, measuring the RNA in the sample, and adding more chemicals to amplify the gene in question. The final product: a measurement of how strongly the target gene for this round of experiments is expressed.
Behind Mohns, on the screen of one of the lab’s computers, a simple graph displays curves like overlapping sine waves, the results of tests run on other samples. A glance at the axes reveals that this graph tracks the activity of lysozyme, one of Gurney-Smith’s 50-odd genes, which codes for a protein related to immune system functioning. Each line measures lysozyme expression from a raft scallop, collected several months apart. Sure enough, each line is different, showing that the gene expression levels have changed with the ocean around them.
The shellfish are finally telling us where it hurts.
* The Hakai Institute and Hakai Magazine are both part of the Tula Foundation. The magazine is editorially independent of the institute and foundation.